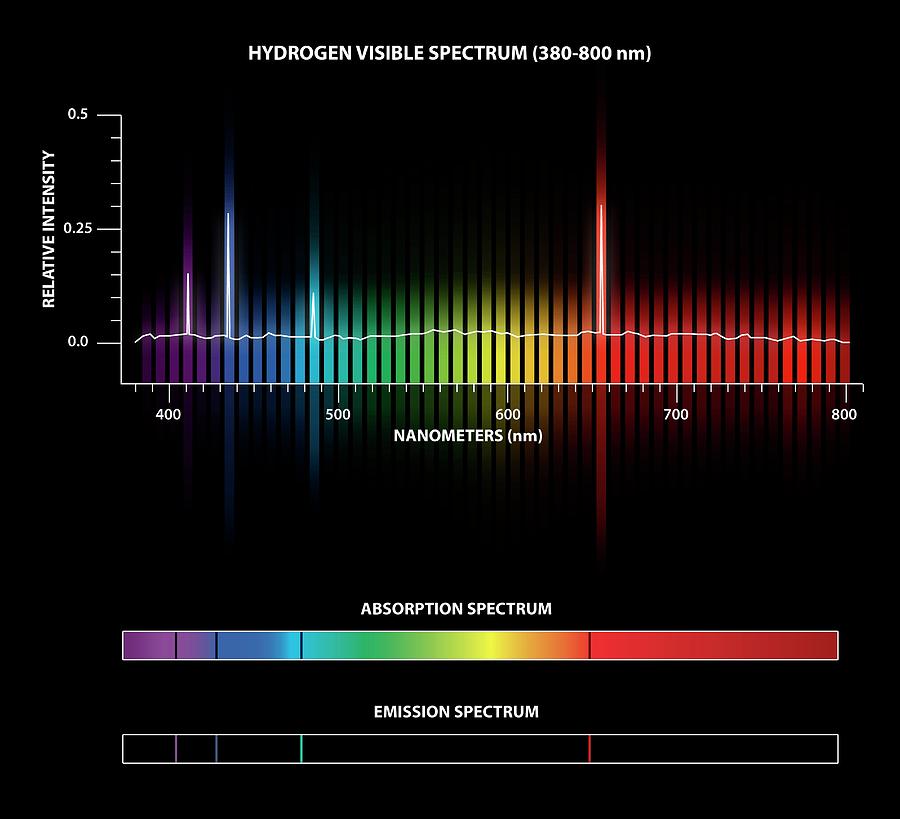
It would tend to lose energy again by falling back down to a lower level. Suppose a particular electron is excited into the third energy level. Hydrogen molecules are first broken up into hydrogen atoms (hence the atomic hydrogen emission spectrum) and electrons are then promoted into higher energy levels. The high voltage in a discharge tube provides that energy. But if energy is supplied to the atom, the electron is excited into a higher energy level, or even removed from the atom altogether. When unexcited, hydrogen's electron is in the first energy level-the level closest to the nucleus. Why does hydrogen emit light when excited by a high voltage and what is the significance of those whole numbers? Each line can be calculated from a combination of simple whole numbers. The lines in the hydrogen emission spectrum form regular patterns and can be represented by a (relatively) simple equation. The origin of the hydrogen emission spectrum The various combinations of numbers that can be substituted into this formula allow the calculation the wavelength of any of the lines in the hydrogen emission spectrum there is close agreement between the wavelengths generated by this formula and those observed in a real spectrum.Ī modified version of the Rydberg equation can be used to calculate the frequency of each of the lines: In other words, if \(n_1\) is, say, 2 then \(n_2\) can be any whole number between 3 and infinity. 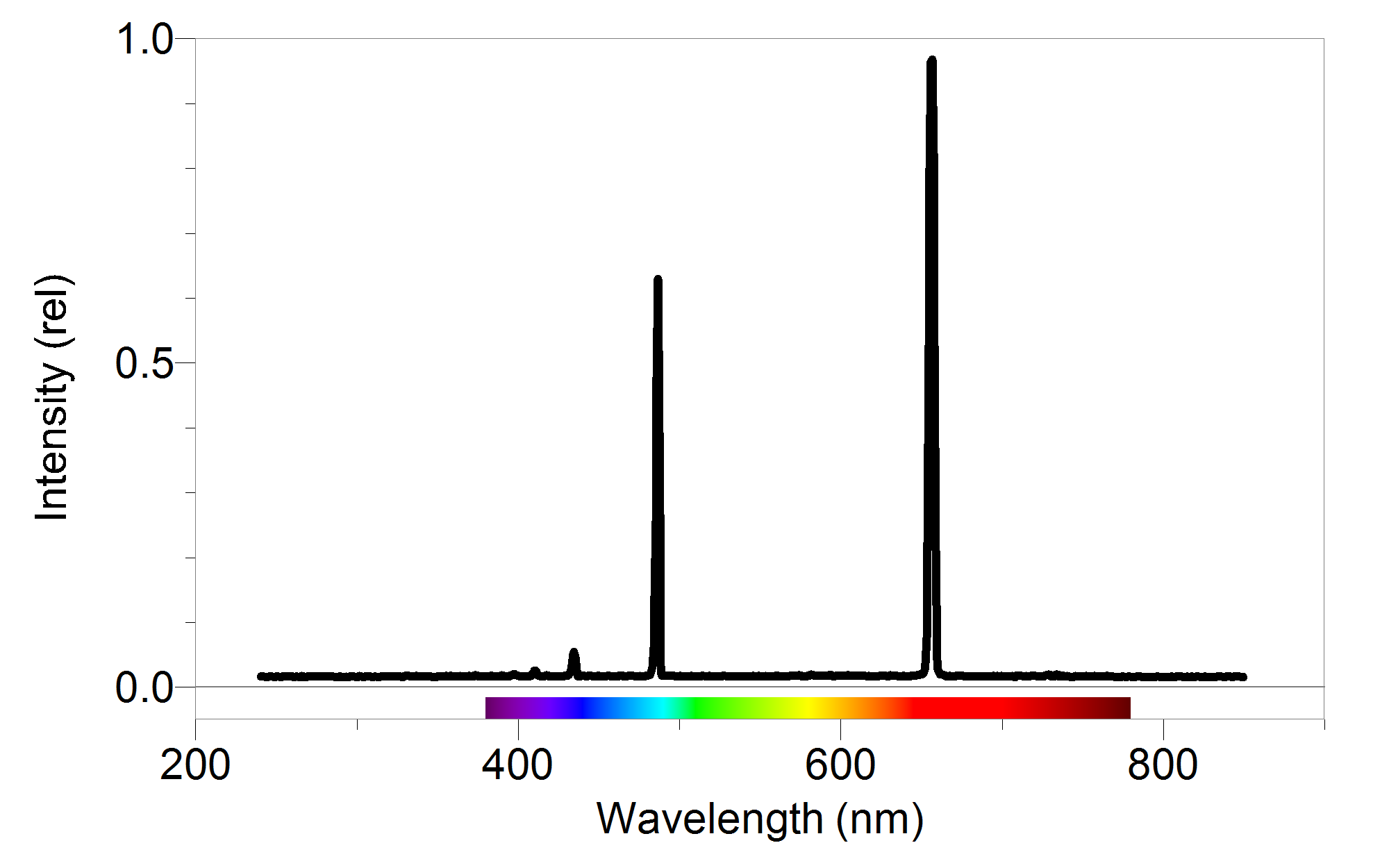
\(n_1\) and \(n_2\) are integers (whole numbers).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
